|
Mendeleev left gaps in his periodic table for yet-to-be-discovered elements, assuming they would fill those spaces.Limitations of Mendeleev’s Periodic Table It’s like guessing a missing ingredient in a recipe and getting it right! Mendeleev’s periodic table laid the foundation for our modern version, making it easier for scientists to understand and work with the diverse world of elements. For instance, there was a gap for germanium, but Mendeleev predicted its existence and even described its properties accurately before its discovery. This helped scientists predict the properties of missing elements. Elements with similar properties lined up in rows and columns. Mendeleev noticed that when he arranged these elements in order of their atomic weights, a pattern emerged. Imagine Mendeleev’s Periodic table like a big, organized grocery list for all the tiny building blocks of matter. In essence, while Newland’s octave didn’t perfectly harmonize, they paved the way for the more precise periodic table we use today. Think of it as trying to create a melody with elements, but scientists later fine-turned the composition with more accurate arrangements. Still, Newland’s contribution laid the groundwork for future developments in understanding the periodicity of elements. However, his idea faced criticism because it didn’t hold true for all elements. 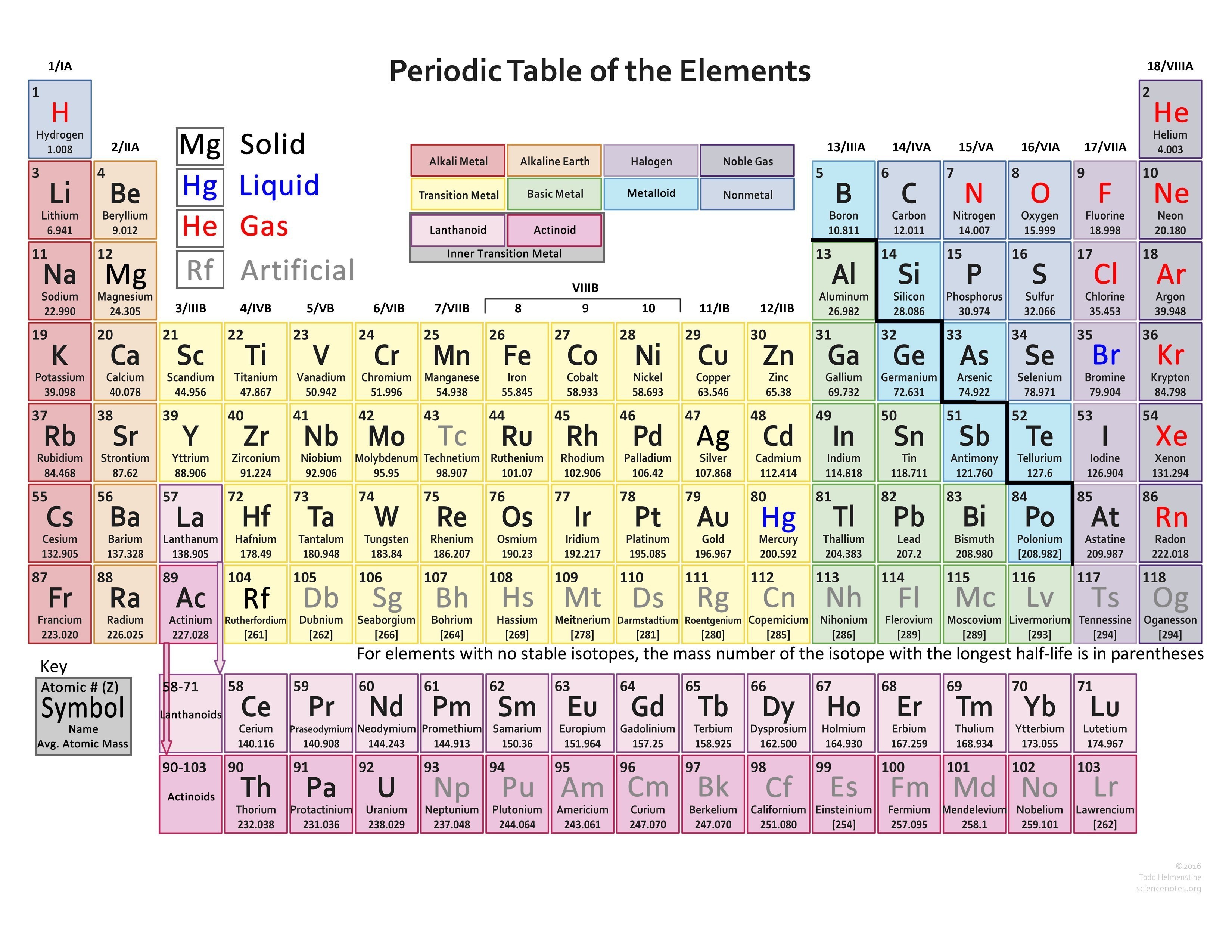
Newlands noticed a repeating pattern in every seventh element, similar to an octave in music. Imagine arranging elements like notes on a musical scale, where every eighth element behaves similarly. Newlands’ octaves were an early attempt to organize elements in the periodic table. (Atomic weight of Li + Atomic weight of K )/2 Despite being a simple idea, Dobereiner’s triads contributed to our understanding of chemical relationships and paved the way for Dmitri Mendeleev’s later development of the periodic table, which organizes elements based on their properties. This concept helped lay the groundwork for the periodic table. For instance, Dobereiner grouped chlorine, bromine, and iodine together, and their atomic masses formed a pattern. In his triads, the middle element had properties that were an average of the other two. He noticed that certain groups of three elements shared similar traits. Johann Dobereiner, a German chemist in the 19th century, had a notable idea called “Triads”. Today, the periodic table is a crucial tool in Chemistry, guiding scientists to predict how different elements will behave and allowing us to unlock the secrets of the elements that make up everything around us. Eventually, they understood that the table’s structure reflected the building blocks of matter. Over time, scientists discovered more elements, and the periodic table grew. He created a table with rows and columns, placing elements with similar traits together. It began in the 1860s when Dmitri Mendeleev, a Russian scientist, noticed that if he arranged elements by their properties, a pattern emerged. The periodic table is like a giant puzzle that helps scientists organize and understand all the elements in the universe. The periodic table helps scientists predict how elements will interact and combines chemistry knowledge into a neat, organized chart that simplifies the complexity of the building blocks of matter. Hydrogen and Helium start things off, and as you move across, elements get heavier. 
The table’s rows are called periods, and represent the energy levels of the elements, while the columns, called groups, highlight shared traits. The periodic table helps scientists predict an element’s characteristics and its reactions with other elements. Each element is represented by a symbol, often derived from its name. 
Elements in the same column share similar chemical behaviors. The Periodic table arranges elements by increasing atomic number, which is the number of photons in an atom’s nucleus. Elements are substances made up of one type of atom. A periodic Table is a chart that organizes all known chemical elements based on their properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
